|
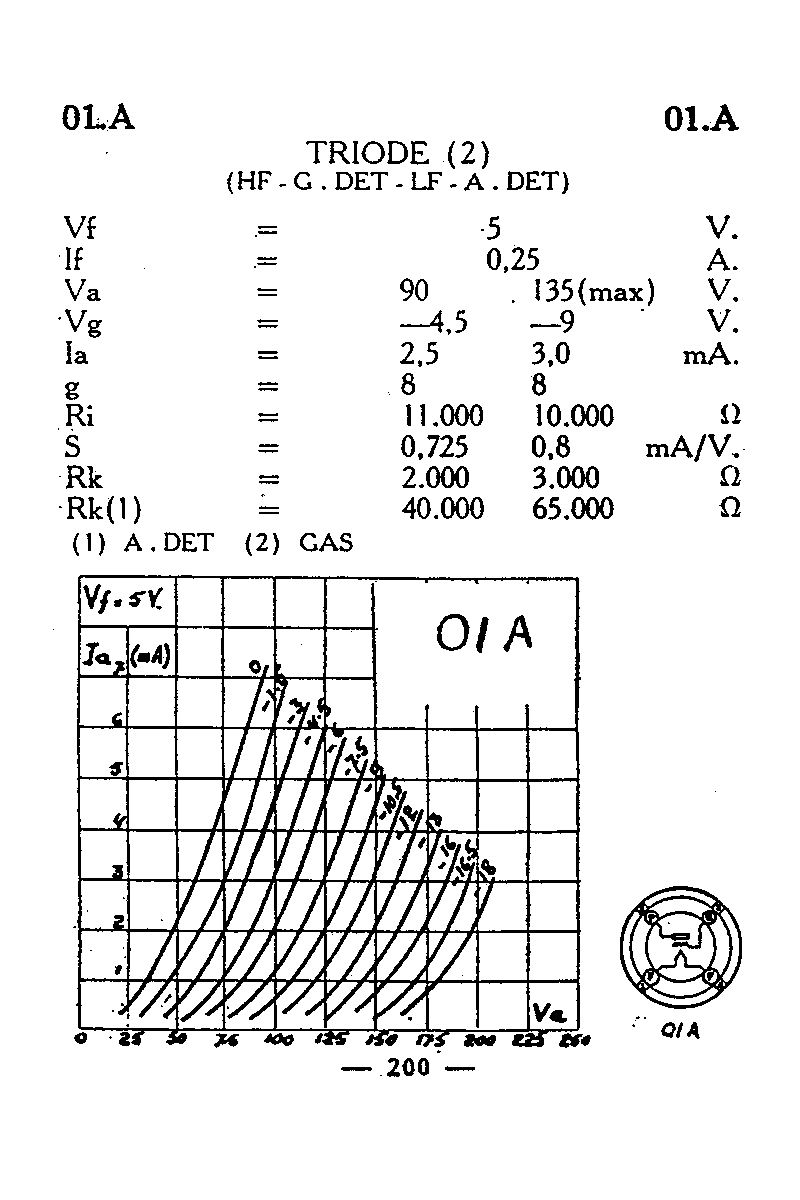
To explain the stabilities, structures, colors, and magnetic properties of transition metal complexes, a different bonding model has been developed. The observed colors indicate that the d orbitals often occur at different energy levels rather than all being degenerate, that is, of equal energy, as are the three p orbitals. 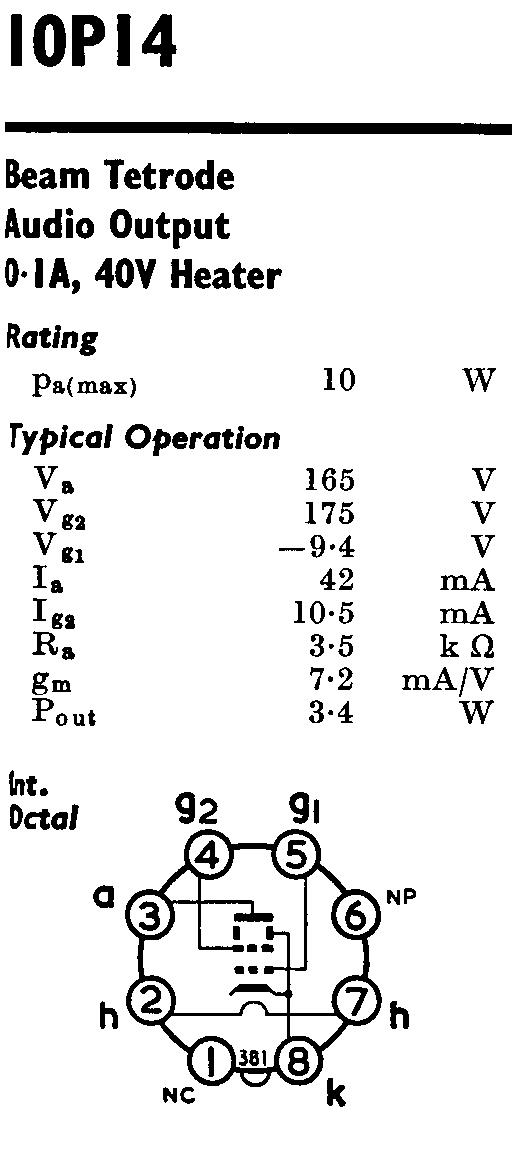
The observed geometries of coordination complexes are not consistent with hybridized orbitals on the central metal overlapping with ligand orbitals, as would be predicted by valence bond theory. The behavior of coordination compounds cannot be adequately explained by the same theories used for main group element chemistry.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
